Bispecific Antibody–Targeted mRNA-LNP Delivery
Efficient delivery of mRNA-lipid nanoparticles (LNPs) to specific cell types remains a major challenge for mRNA cancer vaccines and mRNA immunotherapy. Our centre has developed a generalizable platform that uses bispecific antibodies (BsAbs) to bridge mRNA-LNPs and cell surface markers, enabling precise, targeted mRNA delivery for personalised cancer vaccine applications beyond the liver.
Unlike conventional approaches that modify the lipid composition or functionalize the LNP surface — which complicates mRNA manufacture and alters nanoparticle properties — our BsAb technology leaves the mRNA-LNP unmodified. This innovation enables simplified personalised mRNA production while maintaining efficacy. One arm of the bispecific antibody binds to PEG on the LNP surface, while the other arm carries a custom binder that recognises a protein enriched on the target cell. This bispecific antibody targeting technology was originally developed by Dr Chris Howard and has since been adapted for mRNA cancer vaccine and mRNA immunotherapy delivery by our team.
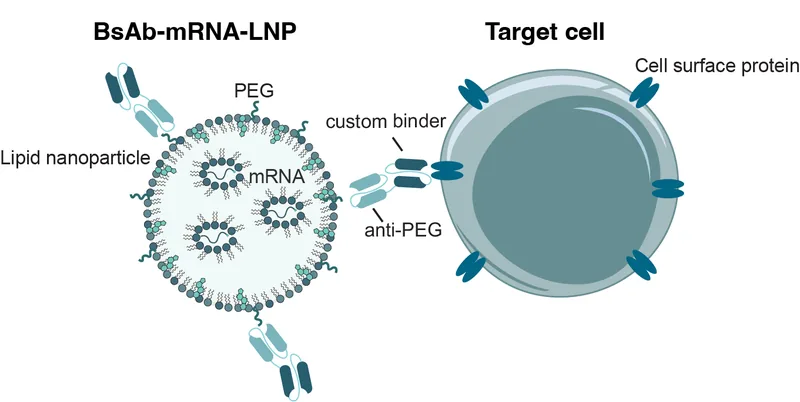
BsAbs form a molecular bridge between mRNA-loaded lipid nanoparticles and target cells.